CPAI’s Research PET/CT Program: 2.9mm Digital Imaging Without Hospital Waitlists

CPAI’s Research PET/CT Program: 2.9mm Digital Imaging Without Hospital Waitlists
Academic researchers and physician investigators face a persistent challenge: securing timely, consistent PET/CT access within hospital systems already overwhelmed by clinical demand.
Study timelines slip. Baseline and follow-up scans happen weeks apart when protocols call for days. Multi-phase trials bog down in scheduling conflicts between research needs and emergency clinical priorities.
Central Park Advanced Imaging operates differently. Our PET/CT program provides dedicated research access on your timeline—not the hospital’s.
Independent Research Infrastructure
CPAI’s United Imaging uMI 550 digital PET/CT operates independently from hospital clinical workflows. No emergency department conflicts. No insurance authorization delays. No waitlists driven by inpatient demand. Just reliable, high-resolution molecular imaging aligned with your research milestones.
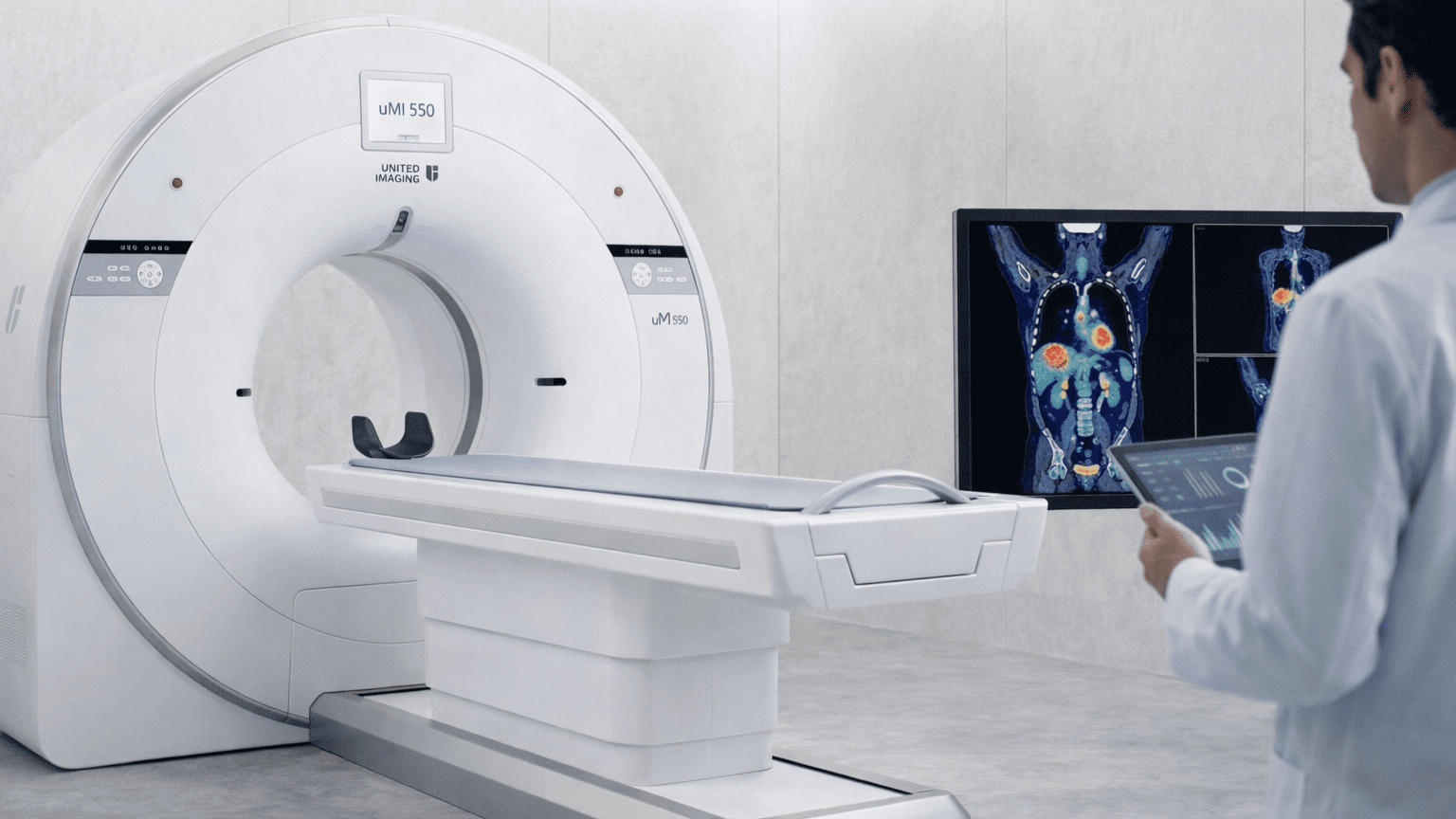
The Platform: United Imaging uMI 550 Digital PET/CT
Research-grade molecular imaging requires more than availability—it demands quantitative consistency, reproducible protocols, and image quality that supports confident analysis.
The uMI 550 delivers:
- 2.9mm Digital PET Resolution: Ultra-fine molecular detail for detecting early-stage disease and small lesions
- 8-Minute Whole-Body Acquisitions: Faster scans improve participant comfort and study completion rates
- AI Motion Correction: Sharper image fusion despite physiological patient movement
- Digital Detector Technology: Improved signal-to-noise ratio compared to analog systems
- Integrated 80-Slice CT: Precise anatomical correlation with metabolic data
This isn’t a clinical PET/CT system retrofitted for research. It’s a digital platform selected specifically for sensitivity, speed, and quantitative performance.
Flexible Scheduling Around Your Study Design
Hospital PET/CT scheduling operates on clinical urgency—cancer staging takes priority over research protocols. Delays between baseline and follow-up scans introduce variability that compromises data quality.
CPAI’s research program schedules around your study milestones:
- Same-day or next-day protocol review and approval
- Imaging timelines structured to match your research plan
- Consistent availability for serial imaging across study phases
- No administrative delays between repeat scans
- Long-term access for pilot studies and multi-year programs
Reproducible Data Across Study Timepoints
Research depends on consistency. When imaging parameters shift between baseline and follow-up, data integrity suffers.
Our platform ensures:
- Standardized acquisition protocols maintained across all study scans
- Quantitative consistency for reliable longitudinal comparison
- Technical quality controls applied to serial imaging
- DICOM export in standardized format for universal compatibility
- Secure data transfer with confidential storage solutions
IRB & Sponsor Ready
Quality-controlled hardware. Board-certified radiologist oversight. Protocol execution by experienced imaging staff. These aren’t optional for research—they’re requirements for IRB approval and sponsor evaluation.
CPAI provides the infrastructure and documentation clinical trials demand.
Applications Across Research Domains
Our digital PET/CT platform supports diverse research applications:
Oncology: Tumor detection, staging, treatment response monitoring, and novel radiotracer development for targeted therapy visualization.
Neurology & Neurodegeneration: Alzheimer’s disease research, Parkinson’s evaluation, amyloid plaque imaging, and epilepsy focus localization.
Infection & Inflammation: Localization of inflammatory conditions, macrophage congregation visualization, and infection source identification.
Call to Action
Academic institutions, physician investigators, and biotech partners: secure reliable PET/CT access for your research. Schedule a consultation or call (212) 363-7315 to discuss your study needs. Visit our Research PET/CT program page for complete details. Contact us for protocol review and access arrangements.
